The energy needs of the humankind are constantly increasing. Satisfying these needs by burning fossil fuels results in pollution, undesired climate changes and eventual exhaustion of the fuel reserves. Nuclear energy is not problem-free either.
Renewable energy is likely to provide the solution for these challenges. The sun offers the most abundant potential source of energy and delivers 3,850,000x1018 J per year to Earth, compared to ~630 x1018 J per year currently consumed by the human civilization. Sun energy was supporting the life on Earth for billions of years via process of photosynthesis. (In fact, all existing fossil fuel reserves originate from photosynthesis processes of long ago.)
As a result of evolution, proteins performing the first steps of photosynthesis achieved amazing effectiveness. Can we learn something useful from Nature by exploring how these first steps are organized? To this end, we explore energy and primary charge separation processes in isolated photosynthetic proteins.


 Top view of plant Photosystem I. Blue: protein. Grey: chlorophylls. Yellow: carotenoids. Red: quinones, Orange: FeS clusters.
Top view of plant Photosystem I. Blue: protein. Grey: chlorophylls. Yellow: carotenoids. Red: quinones, Orange: FeS clusters.
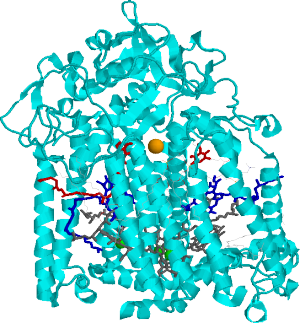 Side view of the bacterial reaction center (BRC). Grey: bacteriochlorophylls. Dark-blue: bacteriopheophytines, Red: quinones, Orange: non-heme iron.
Side view of the bacterial reaction center (BRC). Grey: bacteriochlorophylls. Dark-blue: bacteriopheophytines, Red: quinones, Orange: non-heme iron. 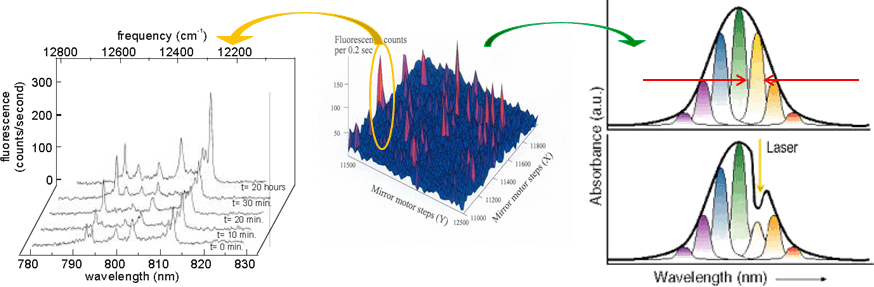 Relationship between single photosynthetic complex spectroscopy and spectral hole burning. The raster-scan image of the sample containing multiple complexes (of the same type, e.g. LH2) is in the center. One can select a single complex (left, one complex contains multiple chlorophyll molecules, hence multiple spectral lines, © Köhler’s Group, Bayreuth), or one can work with the whole ensemble (right) and select molecules in resonance with the narrow-band laser. Then a small conformational change of the environment may be triggered by light, shifting the absorption of these molecules.
Relationship between single photosynthetic complex spectroscopy and spectral hole burning. The raster-scan image of the sample containing multiple complexes (of the same type, e.g. LH2) is in the center. One can select a single complex (left, one complex contains multiple chlorophyll molecules, hence multiple spectral lines, © Köhler’s Group, Bayreuth), or one can work with the whole ensemble (right) and select molecules in resonance with the narrow-band laser. Then a small conformational change of the environment may be triggered by light, shifting the absorption of these molecules.




