Study of the primary processes of photosynthesis
A large family of photosynthetic organisms is capable of the catalytic conversion of the water into molecular oxygen and hydrogen-ions. This process uses inexhaustible resources, such as sunlight, water, and carbon dioxide and provides an example of a unique natural biocatalyst.
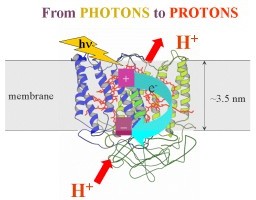
Thorough understanding of the natural solar energy conversion is essential in the process of developing artificial energy converters for sustainable future energy production. The reactions leading to the energy conversion and storage take place in specially organized membrane-bound pigment-protein complexes, termed reaction centers. The energy conversion in these enzymes is secured primarily trough transporting electrons and protons across their natural membranes.
We are particularly interested in the link between the light-induced electron transfer and the accompanying protonational reactions occurring in these centers during the early stage of the energy conversion process. We use an interdisciplinary approach to detect and modify these reactions that can connect concepts from physics, chemistry and biology. We grow and harvest photosynthetic organisms, isolate and purify the reaction center protein. The membrane environment of the isolated proteins is systematically altered in order to maximize the efficiency of the electron and proton transfer reactions.
The biophysical characterization involves transient and steady state optical spectroscopy to determine the kinetics of the individual reaction steps from nanoseconds to minutes time scale and dual polarization interferometry to follow the conformational rearrangement of the protein in real time and in atomic resolution.
Why study photosynthesis? Nature's photosynthetic process has been the primary solar energy conversion on Earth for 3.5 billion years and has a great potential to inspire the development of man-made solar energy converters.



 Group members 2016 (left to right): Chuck Protheroe, Laszlo Kalman and Sasmit Deshmukh
Group members 2016 (left to right): Chuck Protheroe, Laszlo Kalman and Sasmit Deshmukh




