Our interdisciplinary group, situated within both the Department of Physics and Biology at Concordia University, works on the development of an image-guided, targeted drug/gene delivery platform using biomedical ultrasound for the treatment of cardiovascular disease and cancer. This involves using focused ultrasound and acoustically-sensitive agents to spatially and temporally target therapeutic delivery to regions of disease. We use physical, biological, mathematical and engineering-based approaches to address the following major research themes:
- Physics of acoustically-sensitive microbubbles and droplets
- Physical acoustics that initiate safe, transient sonoporation
- Cellular repair mechanisms and their associated time-course
- Key signaling molecules that initiate, sustain and reverse membrane permeability



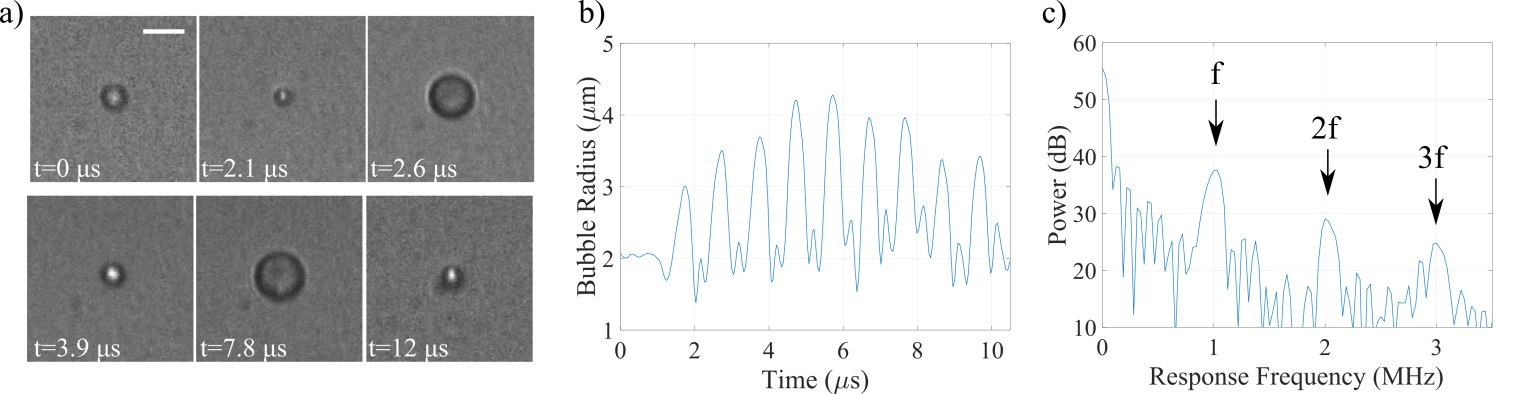 An oscillating microbubble under an ultrasound field (frequency of 1 MHz, peak-negative pressure of 400 kPa, pulse duration of 8 µs) captured at 10.86 million fps. Scale bar is 5 µm. Adapted from Helfield et al. Ultrasound in Medicine and Biology 42(3) 2016. b) Radial cavitation dynamics and c) associated power spectrum. Under such an acoustic regime, this bubble oscillates at the fundamental (f), second (2f) and third (3f) harmonic frequencies, exhibiting distinctly nonlinear behaviour.
An oscillating microbubble under an ultrasound field (frequency of 1 MHz, peak-negative pressure of 400 kPa, pulse duration of 8 µs) captured at 10.86 million fps. Scale bar is 5 µm. Adapted from Helfield et al. Ultrasound in Medicine and Biology 42(3) 2016. b) Radial cavitation dynamics and c) associated power spectrum. Under such an acoustic regime, this bubble oscillates at the fundamental (f), second (2f) and third (3f) harmonic frequencies, exhibiting distinctly nonlinear behaviour.
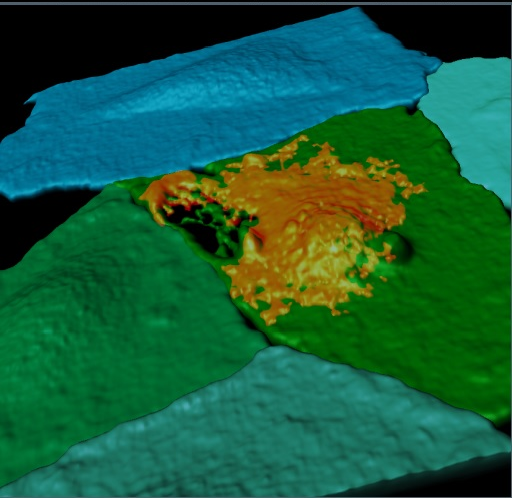 Pictured is a fluorescently labeled endothelial cell monolayer, pseudocolorized in blue/green, and imaged using confocal microscopy. One cell has been selectively perforated via ultrasound-induced microbubble vibration allowing the entrance of a model therapeutic (orange). This perforation re-seals after 30 minutes. See Helfield et al. PNAS 113(36) 2016.
Pictured is a fluorescently labeled endothelial cell monolayer, pseudocolorized in blue/green, and imaged using confocal microscopy. One cell has been selectively perforated via ultrasound-induced microbubble vibration allowing the entrance of a model therapeutic (orange). This perforation re-seals after 30 minutes. See Helfield et al. PNAS 113(36) 2016.





