Naghmeh Bandari received her Bachelor’s and Master’s degrees in biomedical engineering from Amirkabir University of Tehran, Iran, where she served as a Certified Medical Device Technical Supervisor for six years. She is a doctoral candidate of Mechanical Engineering at the Optical-Bio-MEMS and Robotic Surgery Labs. She is also a fellow of Surgical Innovation at the Natural Sciences and Engineering Research Council of Canada (NSERC) CREATE program for Innovation-at-the-Cutting-Edge (ICE). Her research focuses on the development of optical tactile sensors and micro-optical mechanical systems for robotic and surgical applications.
Blog post
Micro-scale technology at war with micro-scale foe
 3D visualization of SARS-CoV-2 virus | Photo by Fusion Animation
3D visualization of SARS-CoV-2 virus | Photo by Fusion Animation
What is COVID-19?
All of us, poor and rich, young and old, men and women, have been cornered by a 0.1-micrometer virus, known as SARS-CoV-2, causing COVID-19 disease. This virus is a member of a family of viruses named the Corona family since they look like a crown under the microscope. Coronaviruses cause infection in our respiratory systems. This infamous family is also responsible for MERS and SARS outbreaks in recent years.
Why timely and accurate diagnosis of COVID-19 is crucial?
Based on the current epidemiologic statistics, a COVID-positive person spreads the virus to 2.3 other individuals (on average) before her/his symptoms surface. The secondary patients also will spread the virus at the same rate before their symptoms are detectable—this process cascades generations of positive cases within only a couple of weeks. If only the first case, even without apparent symptoms, is diagnosed and separated from the society (through self-quarantine or hospitalization), the chain of spreading virus will break very soon. This, however, requires fast and vast testing throughout society.
Other than speed, accuracy is very important in diagnosing COVID-19 cases, whereas, a single COVID-positive patient, if falsely diagnosed as COVID-negative, could start a cascade of spreading the virus.
The current diagnostic technology is accurate, however, doesn’t allow for rapid diagnosis and takes up to days to conclude. But why? and is there a way to decrease the time of test-to-conclusion?
Current technology for the diagnosis of COVID-19?
The core of the current COVID-19 test protocols is the real-time reverse transcription-polymerase chain reaction, or in short, the PCR technique. To detect the presence of the Coronavirus gene in a clinical sample, the PCR technique first amplifies the DNA content of the sample and then uses an optical technique to check for the gene. The latter happens in a split of a second. However, the former (DNA amplification) may take up to days to happen. The reason the DNA amplification is necessary is to increase its content higher than the minimum detectable threshold of the optical detector. So, in short, we need to increase the amount of virus materials to a detectable level.
Now, here is an important question: Can’t we just decrease the size of the detection device so that the relative amount of virus material becomes comparable to the minimum detectable threshold?
New MEMS-based diagnosis of COVID-19
A MEMS is a small device that has mechanical and electronic components. The dimensions of a MEMS range from millimeters to tenth of micrometers. MEMS include components like microsensors, microprocessors and microactuators. They may also have parts to entrap small particles such as viruses.
Researchers have recently shown that the MEMS-based PCR devices and techniques are faster and more accurate than the traditional PCR (find it here). This study showed that the MEMS-based techniques could detect Coronavirus in samples with 500 times lower viral load than conventional PCR.
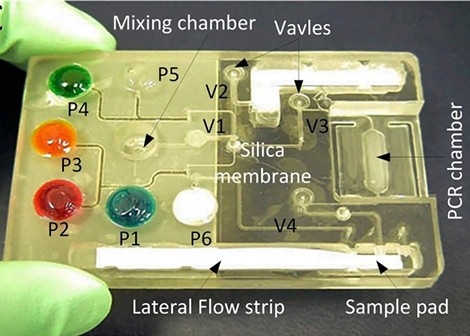
As can be seen in the picture above, a MEMS device integrates multiple laboratory equipment on a single piece smaller than a credit card. This is why some also categorize such a device as a lab-on-a-chip (LOC). LOCs are fast and economical to mass-produce, and easily transportable to urban and rural areas, both making nationwide testing for COVID-19 easy and affordable for governments.
Where are we in the development and mass production of MEMS-based diagnostic devices?
The urgency of the fight against this pandemic has boosted the academic and industrial efforts for developing rapid diagnosis devices and methods. For example:
Stilla Technologies (France) has partnered with Chinese corporations to mass produce Naica digital PCR systems locally in China to help the cities of Xinyang and Zhengzhou. Also, Bosch (Germany) has joined efforts with Randox Laboratories (Ireland) for launching a Vivalytic rapid analyzer. Similarly, in the US, Bio-Rad (California) has partnered with Biodesix (Colorado) to bring Emergency Use Authorization (EUA) COVID-19 test to the US.
 Vivalytic analyzer and test cartridges | Photo by Bosch GmbH
Vivalytic analyzer and test cartridges | Photo by Bosch GmbH
 The MEMS-based diagnostic device developed by the researchers of the University of Cincinnati | Photo by Joseph Fuqua II, UC Creative Services
The MEMS-based diagnostic device developed by the researchers of the University of Cincinnati | Photo by Joseph Fuqua II, UC Creative Services
Final word
With the outbreaks of SARS, MERS, and now COVID-19, rapid test devices will play a significant role in early diagnosis, isolation, and timely treatment of the cases.
The portable devices could also decrease the commute to the hospitals, which decreases the likelihood of spreading the virus and burden on the healthcare facilities
For COVID-19, we are still in the first stage of this pandemic and, unfortunately, but certainly, have more outbreaks coming at us in the future. This is why biomedical researchers and funding agencies must prioritize looking into the potentials MEMS technology and microfluidics to help in the global fight against viral outbreaks.
About the author